News

September 30, 2016
Pacific Oxidants, Sulfur, Ice, Dehydration, and cONvection (POSIDON) Experiment
The NASA Pacific Oxidants, Sulfur, Ice, Dehydration, and cONvection (POSIDON) Experiment is a focused airborne science mission to study the ozone distribution, sulfur chemistry, very short-lived ozone depleting species (VSLS), cloud microphysics, and dehydration in the tropical upper troposphere and lower stratosphere over the western Pacific.July 8, 2016
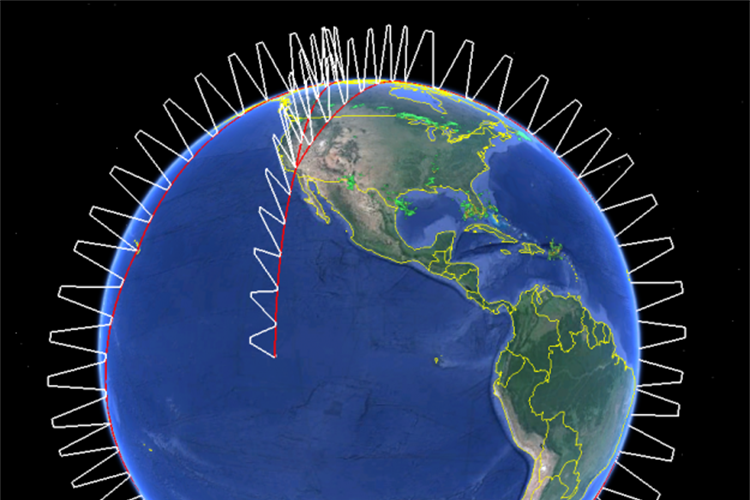
Atmospheric Tomography Mission (ATom)
The Atmospheric Tomography Mission (ATom) is a NASA-funded multi-agency effort using the NASA DC-8 research aircraft to systematically sample trace gases and aerosols from sea level to the stratosphere on 10 pole-to-pole flights covering the Atlantic and Pacific oceans over the next 3 years. ATom will study the impact of human-produced air pollution on greenhouse gases and on chemically reactive gases in the atmosphere with a focus on ozone, methane, and black carbon, as well as atmospheric particulate matter.May 18, 2015
Bottled air from all over the world tells story of ozone-depleting gases and their connection to climate change
If you’re like me, when you hear the word “flask,” you’re likely to picture a grizzled, trail-weary cowboy gulping down a mouthful of whiskey from a tarnished, dented tin. But say “flask” to atmospheric chemist Steve Montzka, and he sees something more like a fire extinguisher or a stainless steel, two-liter soda bottle.May 14, 2015
Recent adjustments to the Montreal Protocol help protect ozone layer, but newer chemicals contribute to warming
An international agreement in 2007 to deal with the last remaining ozone-depleting chemicals used in large quantities is working, according to a new analysis published today. Atmospheric emissions of those chemicals, called hydrochlorofluorocarbons (HCFCs) and used in refrigeration and air conditioning, are no longer increasing, after having increased consistently over the past few decades, according to NOAA measurements published in the Journal of Physical ChemistryJanuary 27, 2014
UAS with NOAA ESRL instruments flies into the Earth’s coldest tropopause.
NOAA ESRL is participating in NASA Global Hawk Unmanned Aircraft System (UAS) research flights from an airbase in Guam to study the coldest parts of the Earth’s tropopause over the tropical Western Pacific.February 1, 2013
Global Hawk UAS Study of Climate Changing Stratospheric Water Vapor & Ozone
The first science flights of the NASA Global Hawk UAS in the winter portion of the Airborne Tropical TRopopause EXperiment (ATTREX) are set to begin the week of 28 January 2013. Six science flights from Edwards Air Force Base, California are scheduled. The UAS experimental payload includes two NOAA/ESRL instruments measuring water vapor, two measuring ozone and one measuring methane, nitrous oxide, hydrogen, and sulfur hexafluoride. Five NOAA/ESRL and six CIRES scientists are at the NASA Dryden facility supporting the missions.September 16, 2011
Unmanned Aircraft Study of Stratospheric Water Vapor & Ozone, and Climate
Five NOAA/ESRL and five CIRES cooperative institute scientists will operate four atmospheric instruments including two ozone sensors, one water vapor sensor, and one greenhouse gases sensor for methane, nitrous oxide, and sulfur hexafluoride on the NASA Global Hawk Unmanned Aircraft Systems (UAS) to study the earth’s Tropical Tropopause LayerAugust 8, 2011
Fifth and Final Pole-to-Pole Aircraft Study of Greenhouse Gases is Underway
Six NOAA and twelve CIRES cooperative institute employees from the NOAA Global Monitoring and Chemical Sciences Divisions of ESRL are involved in the fifth and final HIAPER Pole-to-Pole Aircraft Study of Greenhouse Gases and Black Carbon (HIPPO/5) survey beginning on August 9, 2011, and ending on September 9.January 6, 2011
Fourth Pole-to-Pole Airborne Study of Greenhouse Gases and Black Carbon
The fourth, month-long, HIAPER Pole-to-Pole Observations of Greenhouse Gases and Black Carbon (HIPPO/4) aircraft survey will take place from 14 June to 15 July utilizing 12 flights to cover a total distance of 53,250 km on NCAR’s HIAPER or GV manned aircraft.January 6, 2011
Major Breakthrough by NOAA-led Team on Removal of Air Pollution
An international, NOAA-led research team has made a major breakthrough in understanding the atmosphere’s ability to cleanse itself of air pollutants and some greenhouse gases. Earlier studies were inconclusive on how sensitive the hydroxyl (OH) radical that controls the self-cleaning power of the atmosphere was to environmental changes.